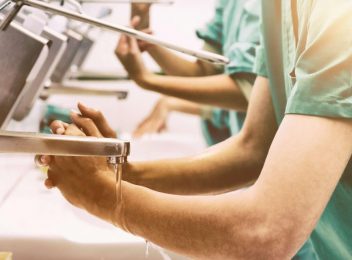
Proteggiamo le persone negli ambienti che le circondano





Azienda e Mission
chi siamo
Nuova Farmec Srl è un’azienda chimico farmaceutica leader italiano nella fornitura di soluzioni per la disinfezione in ambito sanitario dal 1968.
Lottiamo al vostro fianco contro le infezioni in ambito sanitario
Esperienza
Proattività nell’approccio scientifico e conformità all’evoluzione degli stringenti quadri normativi vigenti, contraddistinguono la pluriennale esperienza di Nuova Farmec nella produzione di prodotti disinfettanti e nella prevenzione delle infezioni in ambito sanitario e ospedaliero.
QUALITA'
Lo stabilimento produttivo di Settimo (VR), è un impianto industriale farmaceutico che produce con sistema di qualità certificato in base alle seguenti normative:
a. Conformità alle Norme di Buona Fabbricazione per la produzione di medicinali (GMP)
b. UNI EN ISO 13485
c. UNI EN ISO 9001
SUPPORTO TECNICO
Nuova Farmec dispone di una rete vendite capillarmente distribuita su tutto il territorio italiano in grado di supportare costantemente le esigenze dei propri clienti. È inoltre coadiuvata da un servizio clienti locale e da un team di specialisti e tecnici in grado di fornire assistenza in differenti ambiti nel settore della disinfezione.
SICUREZZA E SOSTENIBILITA'
Nuova Farmec produce e commercializza prodotti di uso consolidato e dalla comprovata efficacia per tutti gli operatori professionali e per i pazienti delle strutture sanitarie in un’ottica di sostenibilità per l’ambiente.